Class 10 Science Lesson 14 Note
#Unit -14 Classification of Elements
1. Choose the correct option for the following questions.
a) In which group do the elements having electronic configuration 1s², 2s² 2p³ lie?
Correct answer: (iii) VA
b) What are the elements between groups IIA and IIIA called in the modern periodic table?
Correct answer: (ii) Transition metals
c) Which one of the given nonmetals is the most reactive?
Correct answer: (i) Fluorine
d) Which one of the given metals is the most reactive metal?
Correct answer: (iv) Cesium
e) Which group do the inert gases belong in the modern periodic table?
Correct answer: (i) 0
f) Which one represents the correct order of increase in chemical reactivity of metals?
Correct answer: (i) Be < Mg < Ca
g) The position of elements A, B. C, and D are shown in the periodic table. Which of them can form acidic oxide?
Correct answer: (iii) C
2. Give reasons:
a) Classification of elements is necessary.
Classification helps in organizing elements systematically, making it easier to study their properties. It also helps predict the behavior and properties of unknown or newly discovered elements.
b) The size of atoms increases on going from top to bottom in a group of the periodic table.
As we move down a group, new electron shells are added, increasing atomic size. Additionally, the effect of increased nuclear charge is shielded by inner electrons.
c) The size of atoms decreases on going from left to right of a period in the periodic table.
Across a period, electrons are added to the same shell while the nuclear charge increases. This stronger attraction pulls electrons closer, reducing atomic size.
d) Hydrogen is a non-metal but it is kept with metals in the modern periodic table.
Hydrogen has one electron like alkali metals, so it shows similar properties in some reactions. Also, it forms positive ions like metals, justifying its placement in group 1.
e) Potassium is more reactive than sodium.
Potassium is larger in size, so its outer electron is more loosely held and easily lost. This makes it more reactive compared to sodium, which has a smaller atomic radius.
f) Fluorine is more reactive than chlorine.
Fluorine is smaller in size, so its nucleus attracts electrons more strongly. It also has a higher electronegativity than chlorine, making it more reactive.
g) The metallic characters of elements decrease and non-metallic characters increase on going from left to right of a period.
As we move right, the ability to lose electrons (metallic character) decreases due to higher nuclear charge. At the same time, elements start gaining electrons more readily, increasing non-metallic character.
h) Inert gases are kept in group 0 of the modern periodic table.
They have completely filled outermost electron shells, making them chemically stable. Because of this, they do not readily form compounds and are called inert.
i) Atomic size decreases from Li to Ne in a period.
Across a period, nuclear charge increases without adding new shells. This pulls electrons closer to the nucleus, reducing atomic size from Li to Ne.
3. Differentiate between:
a) Group vs Period
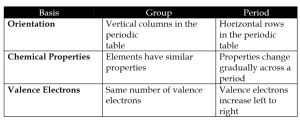
b) Chlorine vs Sodium
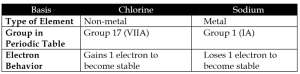
c. Group IA Elements vs Group VIIA Elements
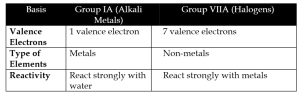
d. Electronegativity vs Electropositivity
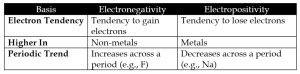
4. Answer the following questions:
a) State modern periodic law.
Answer: The modern periodic law states that:
“The properties of elements are the periodic functions of their atomic numbers.”
b) How many groups and periods are there in the modern periodic table?
Answer: There are 18 groups and 7 periods in the modern periodic table.
c) Write the position of sodium in the modern periodic table. Why is it known as alkali metal?
Answer:
- Group: 1 (IA)
- Period: 3
Sodium is called an alkali metal because it forms strong alkaline solutions (like sodium hydroxide) when it reacts with water.
d) Write the position of the following elements in the periodic table:
Answer:
|
Element Type |
Position in Periodic Table |
|
Alkali metals |
Group 1 |
|
Alkaline earth metals |
Group 2 |
|
Inert gases (Noble gases) |
Group 18 |
|
Transition metals |
Group 3 to 12 |
|
Lanthanides |
Separate row below the table |
|
Actinides |
Separate row below the table |
e) Write the electronic configuration of sulphur and write its position in the modern periodic table.
Answer:
- Electronic Configuration of Sulphur (S): 2, 8, 6
- Group: 16 (VIA)
- Period: 3
f) On which basis are the group and period separated in the modern periodic table?
Answer:
- Groups: Based on the number of valence electrons.
- Periods: Based on the number of electron shells.
g) In which group do Fluorine, Chlorine, and Bromine lie in the periodic table? Which one is more reactive among them?
Answer:
- Fluorine (F), Chlorine (Cl), and Bromine (Br) are placed in Group 17 of the modern periodic table.
- This group is called the Halogen group.
Fluorine is the most reactive among them.
Reason:
- Fluorine has the smallest atomic size among the three.
- It also has the highest electronegativity, meaning it can attract electrons more easily.
- Because of this, Fluorine can quickly gain one electron to complete its outermost shell.
As we move down the group, the size of atoms increases and the reactivity decreases.
So, the order of reactivity is:
Fluorine > Chlorine > Bromine
h) If you are given a chance to make improvements in the modern periodic table, what changes would you make in it? Explain with reasons.
Answer: If I get a chance to improve the modern periodic table, I would:
Place the lanthanides and actinides in the main body of the periodic table instead of keeping them in two separate rows below the table.
Reasons:
- The current table looks disconnected because lanthanides and actinides are shown separately.
- These elements actually belong to periods 6 and 7, but they are not shown in their real positions.
- Putting them in the main table would make the table look complete and easier to understand.
Advantages of this change:
- It will help students to understand the properties and positions of all elements better.
- It will make the periodic table more organized and informative.


