Class 10 Science Lesson 18 Note
#Unit -18 Hydrocarbons and it’s Compounds
1. Choose the best option for the following questions.
a) Which hydrocarbon is included in the alkane group?
i. Methane
ii. Ethene
iii. Acetylene
iv. Propyne
Answer: i. Methane
b) Which one is the correct formula of glycerol?
i. C₃H(OH)
ii. CH(OH)₂
iii. CH₃(OH)
iv. C₃H₅(OH)₃
Answer: iv. C₃H₅(OH)₃
c) Which of the following is the IUPAC name of the alcohol used as a beverage?
i. Methanol
ii. Ethanol
iii. Propanol
iv. Butanol
Answer: ii. Ethanol
d) Which of the following is used to protect skin from dryness?
i. Methyl alcohol
ii. Ethyl alcohol
iii. Glycerol
iv. Glucose
Answer: iii. Glycerol
e) Which hydrocarbon is used to prepare glycerol?
i. Methane
ii. Ethane
iii. Propane
iv. Glucose
Answer: iii. Propane
2. Write differences between:
a) Saturated and Unsaturated Hydrocarbons

b) Alkane and Alkene

c) Monohydric Alcohol and Dihydric Alcohol
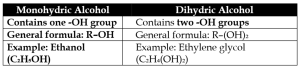
3. Give Reasons
a) Ethane is known as a saturated hydrocarbon.
Reason:
Ethane contains only single bonds between carbon atoms. Since it has no double or triple bonds, it is fully saturated with hydrogen atoms, hence called a saturated hydrocarbon.
b) Glycerol is called a trihydric alcohol.
Reason:
Glycerol contains three hydroxyl (-OH) groups in its molecule. Alcohols having three hydroxyl groups are called trihydric alcohols, so glycerol is classified as one.
4. Answer the following questions.
a) What are hydrocarbons? Write any four examples.
Answer:
Hydrocarbons are organic compounds made up of only carbon and hydrogen atoms. They can be saturated or unsaturated.
Examples:
- Methane (CH₄)
- Ethane (C₂H₆)
- Ethylene (C₂H₄)
- Acetylene (C₂H₂)
b) What do you mean by a saturated hydrocarbon? Write with examples.
Answer:
Saturated hydrocarbons are compounds in which carbon atoms are connected only by single bonds. They are also called alkanes.
Examples:
- Methane (CH₄)
- Ethane (C₂H₆)
- Propane (C₃H₈)
c) Introduce methane gas along with its two uses.
Answer:
Methane (CH₄) is the simplest alkane and a colorless, odorless gas. It is the main component of natural gas.
Uses:
- Used as a fuel in homes and industries.
- Used in the production of hydrogen gas and carbon black.
d) What is alcohol? Write its types on the basis of the hydroxyl group. Also write their examples along with molecular and structural formulas.
Answer:
Alcohols are organic compounds that contain one or more hydroxyl (-OH) groups attached to carbon atoms.
Types of alcohol based on hydroxyl group:


g) Write the structural formula of ethylene. What type of bond is found between its hydrogen and carbon? Why is the bond between its carbon atoms weak?
Answer:
Structural Formula of Ethylene (C₂H₄):
H₂C=CH₂
H H
\ /
C = C
/ \
H H
Bond type between hydrogen and carbon:
- Single covalent bond
Why is the bond between carbon atoms weak?
Answer: The double bond between the carbon atoms consists of one sigma (σ) bond and one pi (π) bond.The pi bond is weaker and more easily broken, making the double bond relatively less stable compared to a single bond.
h) Study the following structural formula of a hydrocarbon and answer the questions.
i. Write the name of this compound.
Answer:
The compound is Ethane.
ii. Is it a saturated hydrocarbon? Write with reason.
Answer:
Yes, ethane is a saturated hydrocarbon because it contains only single bonds between carbon atoms. It has the maximum number of hydrogen atoms attached to the carbon atoms without any double or triple bonds.
iii. Which compound will form if one of the hydrogen atoms is replaced?
Answer:
If one hydrogen atom in ethane is replaced by another atom or group, different compounds can form depending on the substituent. Commonly:
- If a hydrogen atom is replaced by a halogen atom (like chlorine), it forms chloroethane (C₂H₅Cl).
- If replaced by a hydroxyl group (-OH), it forms ethanol (C₂H₅OH).
i) Write any three uses of methane gas.
Answer:
- Used as a fuel in homes and industries for cooking and heating.
- Used as a raw material in the production of hydrogen gas and carbon black.
- Used in the manufacture of synthetic chemicals such as methanol and fertilizers.
j) Write major uses of each of ethane and propane.
Answer:
- Ethane:
Mainly used as a raw material to produce ethylene by cracking, which is a starting compound for making plastics and other chemicals. - Propane:
Used as a fuel for heating, cooking, and in portable stoves. Also used as a fuel in lighters and as a refrigerant.
k) Define the following terms:
i. Saturated hydrocarbon:
Answer: A hydrocarbon in which all carbon atoms are connected by single bonds only; it contains the maximum number of hydrogen atoms possible. Also called alkanes.
ii. Unsaturated hydrocarbon:
Answer: A hydrocarbon that contains one or more double or triple bonds between carbon atoms, meaning it has fewer hydrogen atoms than saturated hydrocarbons.
iii. Alkane:
Answer: A hydrocarbon with only single bonds between carbon atoms, following the general formula CₙH₂ₙ₊₂.
iv. Alkene:
Answer: A hydrocarbon containing at least one carbon-carbon double bond, with the general formula CₙH₂ₙ.
v. Alkyne:
Answer: A hydrocarbon containing at least one carbon-carbon triple bond, with the general formula CₙH₂ₙ₋₂.
vi. Functional group:
Answer: A specific group of atoms within a molecule that is responsible for the characteristic chemical reactions of that molecule. Example: –OH in alcohols.
vii. Homologous series:
Answer: A series of compounds having the same functional group but differing by a CH₂ unit, showing similar chemical properties and a gradual change in physical properties.
l. Write the molecular formula of the following compounds:
Answer:

m) Write the name and structural formula of the alcohol used in spirit lamps.
Answer:
- Name: Methyl alcohol (Methanol)
- Structural Formula: CH3–OH
n) Name the compound formed by the replacement of three hydrogen atoms with three hydroxyl groups (-OH) from propane. Also write its IUPAC name along with its structural formula. Write its three uses.
Answer:
- Name: Glycerol
- IUPAC Name: Propane-1,2,3-triol
- Structural Formula:
HO–CH2–CH (OH)–CH2–OH
Uses:
- Used in cosmetics and skin care products to moisturize skin.
- Used as a solvent in pharmaceutical formulations.
- Used as a plasticizer in manufacturing plastics.
o) Rama has a problem of dry skin in her hands, feet, and face. Which compound can be used to solve her problem? Write the IUPAC name and the structural formula of that compound.
Answer:
- Compound: Glycerol (used as a moisturizer)
- IUPAC Name: Propane-1,2,3-triol
- Structural Formula: HO–CH2–CH (OH)–CH2–OH


